Breakthrough Analyzer (BTA)
A Compact, Versatile, High-Performance Selective Adsorption System.
- ▪ Superior design minimizes dead volume & delivers accurate, experimental results
- ▪ Configurable with up to 6 precision mass flow controllers and 2 vapor sources
- ▪ Patented high-performance blending valves
- ▪ Sample activation up to 1050℃
- ▪ Thermostated environmental chamber provides uniform temperature control, even when using vapors
- ▪ Easily connects to commercial Mass Spectrometer (MS) & Fourier Transform Infrared Analyzer (FTIR)
- ▪ Secure door lock system for enhanced operator safety
General
The new BreakThrough Analyzer (BTA) is a flexible gas delivery and management system for the precise characterization of adsorbent performance under process-relevant conditions. It delivers reliable adsorption data for gas/vapor mixtures using a flow-through system.
A safe and highly optimized device for collecting both transient and equilibrium adsorption data for multi component systems. The BTA can be configured with up to six precision mass flow controllers and patented high performance blending valves, delivering unparalleled flexibility in experimental design. The superior gas-delivery design ensures the precise control of both composition and flow rate, while minimizing dead volume.
The high-quality, stainless-steel column can hold 0.05 to 2.5 grams of adsorbent. Automated sample activation up to 1050°C is possible with the precise, rugged, and reliable resistance furnace.
Operating pressures are controlled from atmospheric to 30 bar via a servo positioned controlled valve. The thermostated environmental chamber delivers uniform temperature control of the entire system up to 200°C, eliminating cold spots. The BTA secure door lock system ensures operator safety throughout the analysis.
Vapor generators can be added to the BTA to enable the use of important probe molecules such as water for experimental studies. The BTA easily connects to commercially available Fourier Transform Infrared and Mass Spectrometer systems for gas identification and quantification.
Breakthrough Adsorption Theory
Breakthrough analysis is a powerful technique for determining the sorption capacity of an adsorbent under flow conditions. Dynamic breakthrough adsorption provides many advantages over static adsorption measurements.
Easily collect multicomponent adsorption data
Determine adsorbate selectivity
Replicate process conditions
When conducting breakthrough analysis, sample preparation is a critical step in the analysis process to prevent pressure drop and mass transfer limitations. Pressure drop occurs when the interstitial space between particles is too small to accommodate the flow rate of gas. Mass transfer limitations occur when the pore size of the material is similar to the kinetic diameter of the adsorbate. Appropriately sizing particles is therefore critical to obtain the best results.
Mass Spectrometer (MS)
Multicomponent adsorption studies often require a mass spectrometer (MS) to monitor the residual gas composition. The MS is the most common detector system used for breakthrough analysis.
FTIR Analyzer (FTIR)
FTIR spectrometers are often selected for experimental breakthrough studies such as the separation of xylenes or other aromatic hydrocarbons.
Humidity Sensor
Allows direct tracking of water content for low cost. Can be useful especially in production control applications.
Sample Preperation System
Small quantities of active material can be mixed with an inert carrier to produce a homogeneous sample and improve analysis reproducibility.
CO2 Sensor
Allows direct tracking of CO2 content for low cost. Can be useful especially in production control applications.
Sample Column (Different Volume)
The BTA may be used with a variety of column diameters to accommodate different sample morphologies included powders, pellets, and extrudates.
MFC and Mixing Valves (Maximum of 6 Gas Inlets)
Additional mass flow controllers and blending valves may be added to the BTA to increase the analytical capabilities and expand the range of experiments that may be conducted.
Vapor Source (Max of 2)
Moisture or other vapors such as xylenes or other aromatics are compatible with the optional vapor sources available for the BTA.

Direct Air Capture
DAC is difficult due to low concentrations of carbon dioxide in air along with other impurities including moisture, and the captured CO2 may be sequestered underground, sold, or converted into value added chemicals to offset carbon emissions.
CO2 Adsorption
Power generation, chemical plants, and refineries are significant point sources for carbon dioxide emissions and the higher concentrations often require different operating conditions when compared to direct air capture

Olefin / Paraffin Separators
Are a core part of the petrochemical industry and used to in the production of polymers such as polyethylene and polypropylene; these separations are energy intensive and increase CO2 emissions.

Natural Gas Separation
Natural gas is a mixture of hydrocarbons and other gases that must be purified prior to use in industrial applications and households for heating and food preparation.
Toxic Gas Adsorption
Porous solids are used for personal protection and also under development for the capture of toxic gases including sulfur dioxide, hydrogen sulfide, and nitrogen dioxide from natural gas or other process feeds.

Water Adsorption
Harvesting water from the air may be a critical technology for many parts of the world clean, where the fresh water supply is limited due to an arid climate or the increasing usage of water for agriculture.

Zeolites
Pressure swing adsorption using Zeolite 5A, 13X, or LiX, which have high selectivity for adsorbing nitrogen are used commercially for air separation and producing oxygen.

Silicas
Amine functionalized silicas are effective and highly selective adsorbents and used for the direct air capture (DAC) of CO2.

Porous Membrane/Monoliths
Porous membranes and monoliths coated zeolites or MOFs are commonly used to improve the operational efficiency of separation processes.

Activated Carbon
Volatile organic component (VOC) from automobile fuel systems are captured by canisters filled activated carbon and these VOC emissions are minimized.

Porous Aluminas
Alumina – Supported Ionic Liquids are effective adsorbents with potential applications for the separation of CO2 from natural gas.

Metal-Organic Frameworks
MOFs are highly selective adsorbents which are effective for demanding commercial applications including alkanes & olefins, olefins & alkynes, DAC and CO2 & CH4.
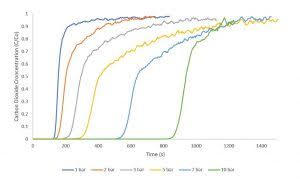
Zeolite 13X has been extensively studied for applications in catalysis and adsorption. In this study, zeolite 13X was used as an adsorbent for carbon dioxide adsorption to collect breakthrough curves from 1 – 10 bar pressure. These measurements were collected using equimolar flowrates of 10 sccm nitrogen and 10 sccm carbon dioxide. A 1 sccm stream of helium was used as a tracer gas to determine the start of the breakthrough experiment. All measurements were collected at an analysis temperature of 30°C. Between each measurement, the zeolite 13X sample was reactivated overnight to ensure complete desorption of carbon dioxide. The figure shows a consistent increase in breakthorugh time across successive experiments as the pressure is increased.
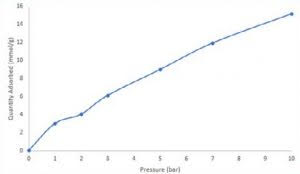
Following carbon dioxide breakthrough measurements an equilibrium adsorption quantity was calculated for each curve by solving the breakthrough equation. Next, an isotherm was constructed displaying the quantity of carbon dioxide adsorbed at 1, 2, 3, 5, 7, and 10 bar total pressure. At 10 bar, zeolite 13X adsorbed roughly 15 mmol/g carbon dioxide. While isothermal data collected via breakthrough cannot be directly correlated with static adsorption measurements, it can provide an assessment of an adsorbent in process-relevant conditions.
Single component carbon dioxide breakthrough adsorption experiments were conducted on zeolites 13X and 5A, and metal-organic frameworks MIL-53(Al) and Fe-BTC. All materials were analyzed at 30°C while flowing an equimolar gas stream consisting of 10 sccm nitrogen and 10 sccm carbon dioxide. A 1 sccm stream of helium was also blended into the feed gas stream as a tracer gas to aid in identifying the start of the breakthrough experiment. The breakthrough curves for the four materials are plotted below on a mass normalized axis. The total quantity of CO2 adsorbed follows the trend: molecular sieve 5A > zeolite 13X > Fe-BTC > MIL-53(Al). The table below shows the total quantity adsorbed in mmol/g.
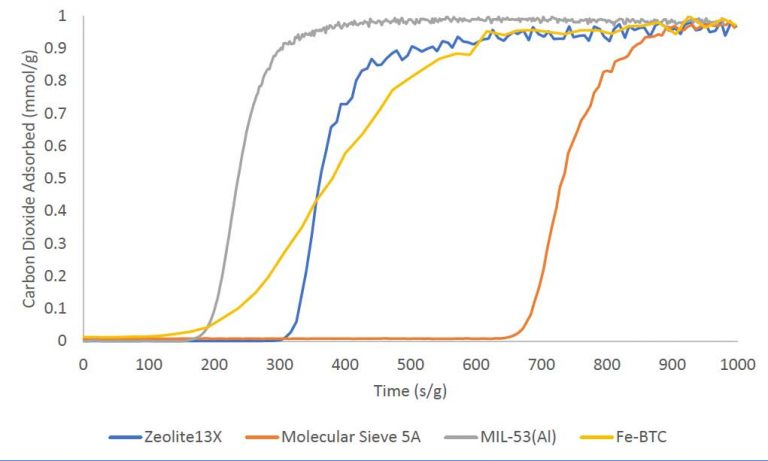
MATERIAL CARBON DIOXIDE ADSORBED(MMOL/G)
ZEOLITE 13X 2.94
MOLECULAR SIEVE 5A 3.52
MIL-53 (AI) 1.23
FE-BTC 2.30
MicroActive the most intuitive, flexible, and comprehensive analysis software for adsorption studies
The flexible, intuitive, easy-to-use software allows for the widest range of experimental conditions and automates the breakthrough from sample activation to sample analysis, including the ability to perform cyclic experiments. Paired with industry leading MicroActive analysis software, the BTA system accurately and reproducibly characterizes adsorbents, analyzes data with comprehensive analysis methods, and solves the breakthrough equation for the most demanding samples.
MicroActive Software allows us:
Data reduction from MS
Quantity adsorbed selectivity
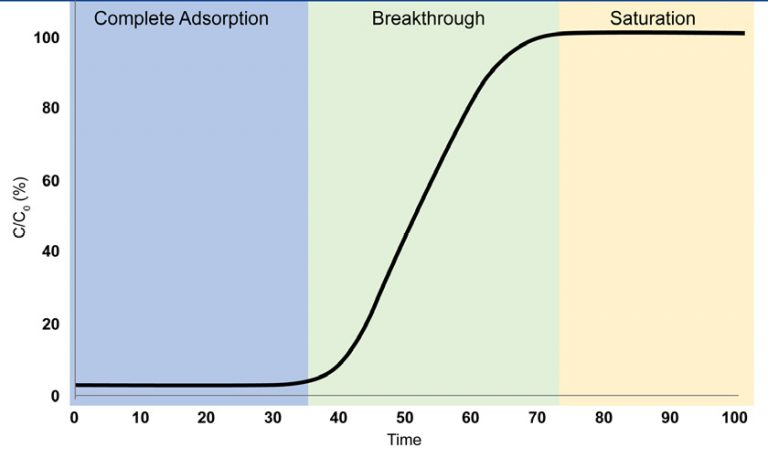
Complete Adsorption
The adsorbate gas is completely adsorbed such that none can be detected at the outlet of the breakthrough column
Breakthrough
The adsorbate gas is first detected at the outlet of the breakthrough column. Gas continues to adsorb; however, the adsorbent is no longer able to adsorb the entirety of the gas that is entering the breakthrough column
Saturation
The adsorbent has reached saturation and can no longer adsorb the adsorbate gas, allowing it to pass through the column freely